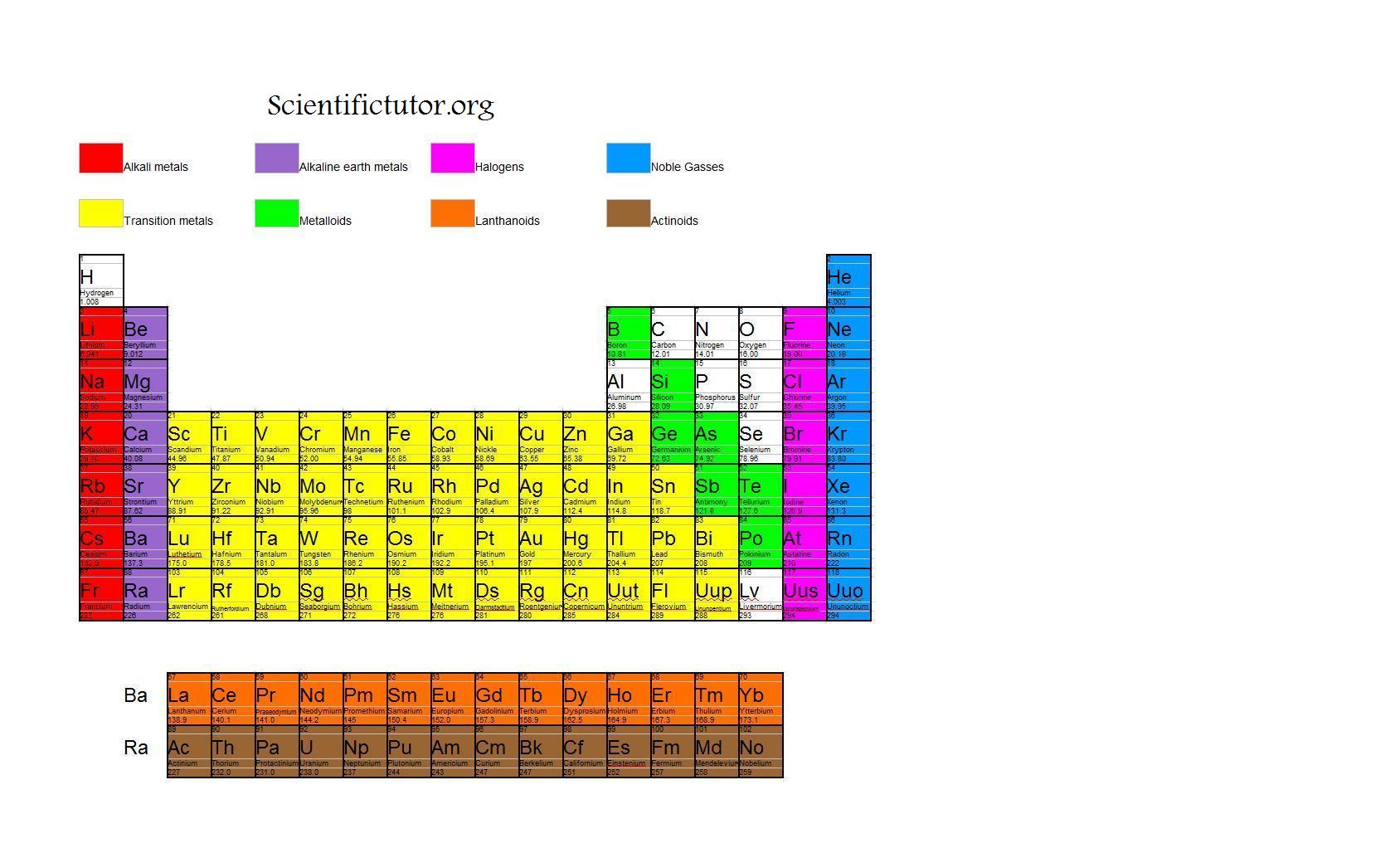
Ionization enthalpy can be defined as the amount of energy required to eliminate the loosely bound electron from an isolated gaseous atom or molecule. The increase in atomic mass and decrease in atomic radius leads to significant increase in atomic density. This trend is observed within elements of a given series. It consequently results in a contraction in size of the element. The reason behind this occurrence is that the d-orbitals commonly serve poor shielding effects, leading to the nuclear charge attracting all the electrons strongly. Generally, the atomic radii of transition elements belonging in the 3d-series are seen to decrease in radius with an increase in atomic number. It follows an exception in the case of Chromium and Copper, where an electron from the 4s shell takes entrance to the 3d shell. The electronic configurations that are involved in elements like Zn, Cd and Hg are usually denoted by the general formula (n-1) d 10ns 2, which is to say that the orbitals of these elements are generally filled in their ground states, alongside their common oxidation states as well. The very first row of transition metals depicts similar electronic configuration. The half or completely filled orbitals are generally more stable. The (n–1) denotes the inner d orbitals that may have anything from between 1 to 10 electrons, while outermost ns orbitals can usually have 1 or 2 electrons in general. The general electronic configuration of transition elements is represented by (n-1) d 1–10 ns 1–2. The d-block comprises 3 transitions series which are formed due to acceptance of electrons by the penultimate energy levels of the atoms.Ĭheck Important Notes for Electron Gain Enthalpy Group 3 - Group 12 elements are the transition elements. The transition elements are commonly located between the ‘s’ and ‘p’ block elements in the modern periodic table, or, in other words, the d-block occupies the largest middle section of the modern periodic table. Position of Transition Metals in Modern Periodic Table Since, Zinc, Cadmium, and Mercury of group 12 possess completely filled d orbitals, they are not referred to as transition metals. Such elements have partially filled or incompleted orbitals either in-ground state or excited state. Pure tungsten was eventually isolated in 1783 by brothers Juan and Fausto Elhuyar.Transition metals are known as d-block elements and are categorized into 3 series, namely, 3d series (Sc to Zn), 4d series (Y to Cd) and 5d series (La to Hg, except Ce to Lu). Most German chemists still refer to the element as Wolfram (wolf's metal). The symbol has its origins in the discovery of the element in the late 1700's linked to the mineral wolframite. Chemistry of Tungsten The name for the metal tungsten is taken from the Swedish, tungsten, for "heavy stone".The longest lived isotope (which has no known practical uses) has a half-life of 800 milliseconds. The American group used a californium target and ions of oxygen-18. The Soviet claim was based on bombardment of three different isotopes of lead with high energy chromium-54 ions. Three months later the Berkeley group claimed positive discovery of the same element. Chemistry of Seaborgium The synthesis of element 106 was first reported by Soviet scientists in June of 1974.The pure metal was first isolated in 1781 by Peter Jacob Hjelm. The metal takes its name from the Greek word molybdos, meaning "lead" because some of its compounds were once confused with similar lead compounds before Scheele's discovery. Chemistry of Molybdenum The first significant practical use of this metal (discovered in 1778 by Scheele) was in the alloy used to manufacture the German artillery gun known to the Allies as Big Bertha.Chromium is also a very useful industrial metal. This element produces many beautifully colored compounds, as well as a wide array of colored solutions. It is named after the Greek word "chroma," meaning color. Chemistry of Chromium Chromium is the 24th element in the periodic table and it is found in about 0.0122% of the Earth's crust.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
